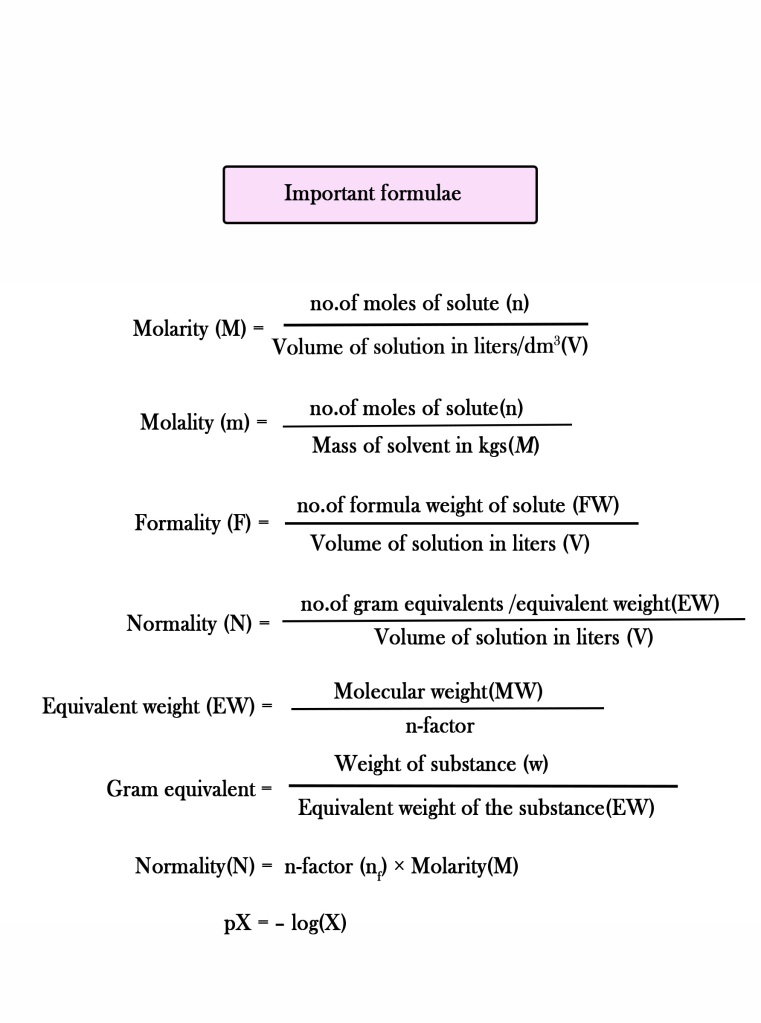
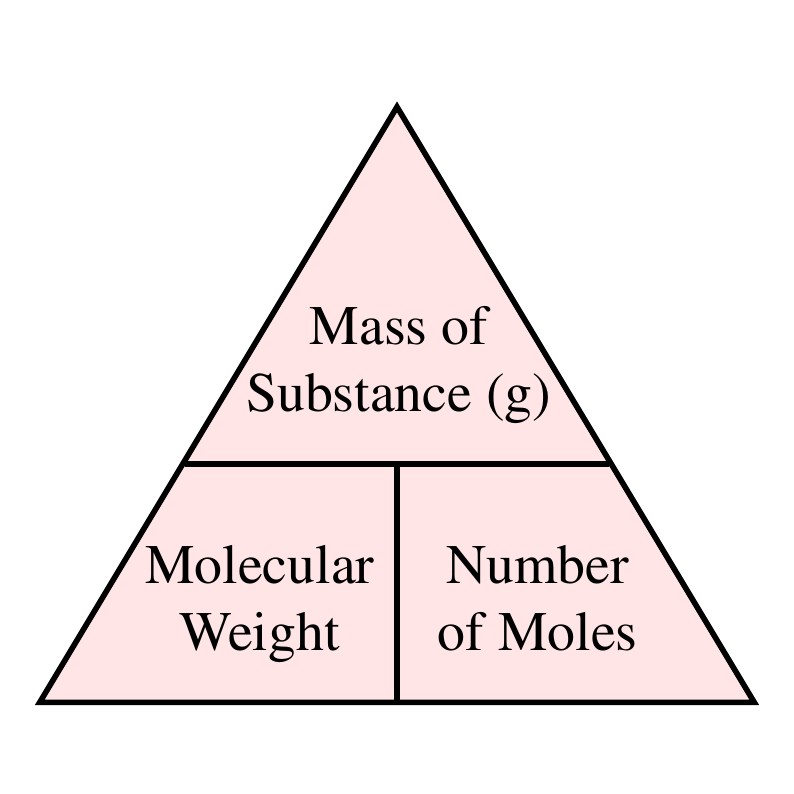
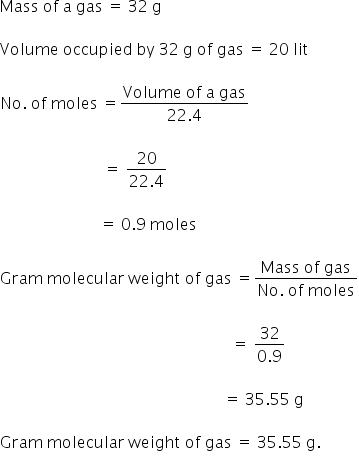Calculate the mass of compound (molar mass = 256 g mol^-1 ) to be dissolved in 75g of benzene to lower its freezing point by 0.48 K (Kf = 5.12 K kg mol^-1 ) .

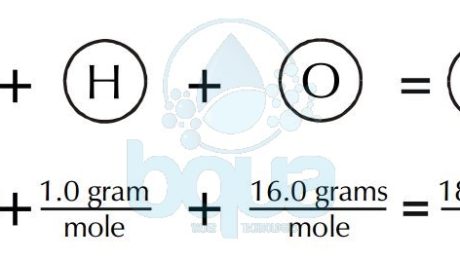
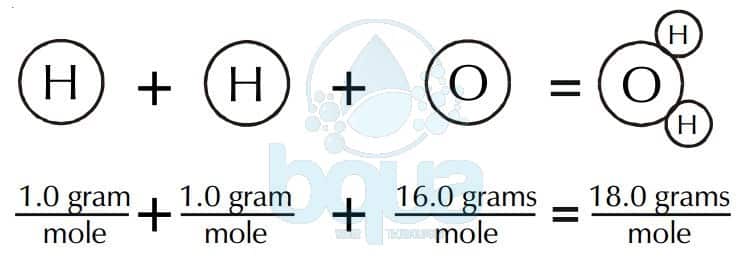
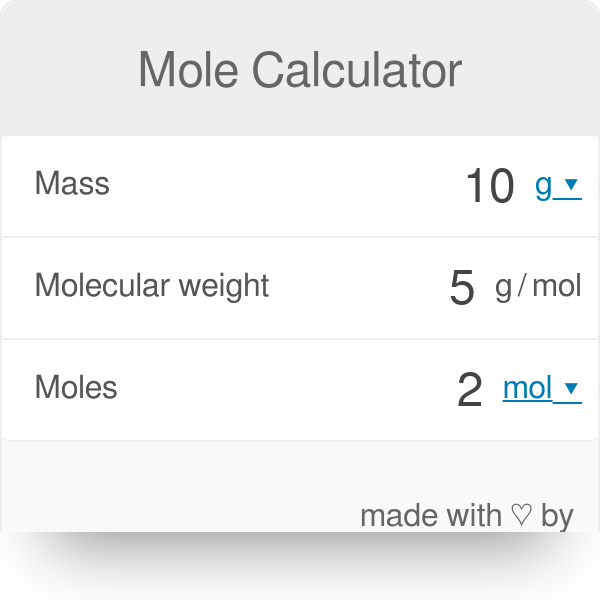
Calculate the gram molecular mass of water from the values of gram atomic mass of hydrogen and of oxygen.Gram atomic mass of hydrogen = 1 g.Gram atomic mass of oxygen = 16 g.

Calculate the gram molecular mass of calcium carbonate involved in this reaction CaCO3 --->CaO - Brainly.in